Research
We use in vivo and in vitro models of cerebrovascular injury and a range of techniques such as in situ hybridization, immunoprecipitation, immunochemistry, high-throughput sequencing, microarrays, reporter assays, gain- or loss-of-function strategies, imaging, and bioinformatics to study RNA biology and function in brain damage.
Focus Area 1: Cerebral ischemia (stroke)
Ischemic stroke is a leading cause of disability and death in the United States. To date, there are no FDA-approved therapeutic interventions available. Our goal is to improve our understanding of the molecular processes driving the cellular and systemic changes that result in brain damage and neurological dysfunction so that we may harness this knowledge to develop new treatments against stroke. Our research is focused on transcriptomics, epigenetics, and gene regulation, and we study how the interplay between RNAs, regulatory proteins, and DNA modulates the gene regulatory networks that underlie the development of the post-stroke pathophysiology. Because RNAs are amenable to therapeutic manipulations, we hope to discover new druggable targets that can be developed for clinical applications against stroke.
Focus Area 2: Chronic cerebral hypoperfusion (CCH) and vascular dementia
Chronic cerebral hypoperfusion (CCH) is a contributing factor to the development of vascular dementia/vascular cognitive impairment, the second most common form of dementia after Alzheimer’s disease (AD). Aging- associated hypoperfusion is widely recognized in the elderly population, however, the pathophysiological link between hypoperfusion and vascular dementia remains poorly understood. Filling this gap in knowledge is of great significance in our quest to halt the development and progression of AD and AD-related dementias (ADRD). Leveraging our expertise in RNA biology, epigenetics, and cerebrovascular injury, our efforts are focused on understanding the relationships between genome organization, gene regulation, gene expression, and cellular outcomes in the cortical and subcortical regions of the brain that are the most significantly affected and implicated in the development of vascular dementia. These findings will be linked to cognitive and behavioral deficits, and novel molecular targets will be identified for therapeutic manipulation to minimize or reverse the adverse behavioral outcomes.
PROJECTS
Project 1: Regulation of gene expression by enhancer-RNAs in cerebrovascular injury
Enhancers undergo activity-dependent transcription to produce noncoding enhancer RNAs (eRNAs). Some of these eRNAs have been shown to play central roles in organizing functional interactions between the enhancers and their downstream gene targets and influence cellular outcomes. We recently identified several novel stroke-responsive eRNAs in the post-stroke cerebral cortex. Loss-of-function experiments resulted in pronounced molecular and phenotypic outcomes, which suggest important roles for the eRNAs in the post-stroke brain. Our work in this area is focused on identifying the molecular targets of these eRNAs, the cellular and physiological processes that they influence, and their sex-based expression and functional characteristics using in vitro and in vivo models of stroke. We are further extending this work to mouse models of vascular dementia to understand the roles of eRNAs in modulating gene expression networks in the subcortical regions of the brain during CCH.
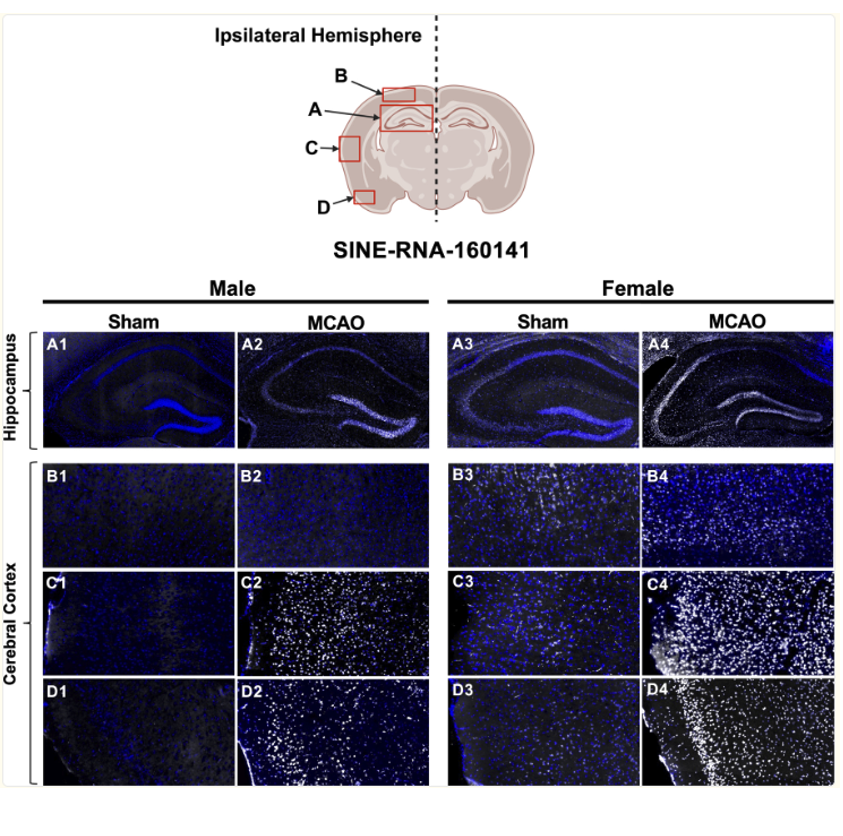
Project 2: SINE-RNAs and post-ischemic gene regulation
We recently identified rapid, RNA polymerase III–driven induction of short interspersed nuclear element RNAs (SINE-RNAs) following cerebral ischemia, revealing a previously unrecognized transcriptional response during acute reperfusion. These RNAs are expressed in neurons across ischemic brain regions, in many instances exclusively in response to ischemia, and are conserved across in vivo and in vitro models. Their expression patterns and preliminary functional data in our lab indicate that they have neuroprotective functions and their nuclear localization suggests roles in chromatin and transcription regulation. Ongoing work in our lab is focused on defining the regulatory mechanisms, cellular functions, and pathological relevance of SINE-RNAs in post-stroke neuroprotection and pathophysiology.
Project 3: Gene expression and brain pathology during CCH
One of the key drivers of vascular dementia and Alzheimer's Disease-related dementia is CCH. CCH takes years to manifest into full-fledged dementia and during that time a number of changes occur at the molecular and cellular level in the brain leading up to overt neurological symptoms. Our lab investigates the mechanisms by which CCH drives vascular dementia/cognitive impairment using the bilateral carotid artery stenosis (BCAS) mouse model. We are characterizing the pathological, cellular, and transcriptional responses to sustained reductions in cerebral blood flow from the early stages of CCH through the late stages of CCH when neurological symptoms are fully manifested. By taking this longitudinal approach, we aim to track the molecular and cellular changes across all stages of the disease and link them to the pathophysiological and neurological outcomes along the trajectory of disease progression. Our work addresses a critical gap in the field by examining sex-dependent responses to CCH, including changes in gene expression, glial activation, cell death, white matter integrity, microinfarct development, and cognitive and sensorimotor functions, with the goal of understanding which features underlie differential outcomes in males and females. Our overarching goal is to identify sex-dependent mechanisms of vulnerability and resilience to CCH that may help identify opportunities for early therapeutic interventions against the development and progression of vascular dementia.